Polysaccharides are complex carbohydrates composed of ten or more monosaccharide units linked by glycosidic bonds. These polymeric polysaccharides have colloidal molecules and serve as essential macromolecules in all living forms. Is starch a polysaccharide? Yes, along with cellulose and glycogen, starch is one of the most important naturally occurring polysaccharides.
Polysaccharides are also known as “glycans.” They can be considered condensation polymers where monosaccharides (or their derivatives, such as amino sugars and uronic acids) are joined by glycosidic linkages.

The classification of polysaccharides divides them into two major types based on their monosaccharide composition:
Common polysaccharides include:
- Homopolysaccharides (Homoglycans): Made up of a single type of monosaccharide. Examples of homopolysaccharides include starch, cellulose, glycogen, and inulin.
- Heteropolysaccharides (Heteroglycans): Composed of two or more different kinds of monosaccharides or their derivatives, such as glycosaminoglycans and peptidoglycan.
Specific polysaccharide nomenclature:
- Polysaccharides made with glucose are called “glucans”
- Polysaccharides made with fructose are called “fructans”
- Polysaccharides made with mannose are called “mannans”
- Polysaccharides made with xylose are called “xylans”
What is the classification of polysaccharides?
The polysaccharide classification system divides these complex carbohydrates based on their monosaccharide composition.
- Homopolysaccharides are polysaccharides formed by the same type of monosaccharides. Classic examples of polysaccharides in this category include starch, glycogen, and cellulose—each formed by hundreds of glucose molecules linked by glycosidic linkages. Is cellulose a polysaccharide? Yes, cellulose is a homopolysaccharide and the most abundant polysaccharide in nature.
- Heteropolysaccharides are polysaccharides whose molecules are formed by different types of monosaccharides. Hyaluronic acid, formed by thousands of alternating units of N-acetyl glucosamine and glucuronic acid, is an example of a heteropolysaccharide with important structural functions in the extracellular matrix.
Homopolysaccharides: Structure and Examples
Homopolysaccharide examples include the three most important polysaccharides for life: cellulose, starch, and glycogen. All three are polymers of glucose but differ significantly in their structure of polysaccharides and biological functions.
a. Cellulose: The Most Abundant Polysaccharide
Is cellulose a polysaccharide? Yes, cellulose is a structural polysaccharide and the most abundant carbohydrate in nature. The general formula of cellulose is (C₆H₁₀O₅)ₙ. Wood contains 50% cellulose, while cotton wool is almost pure cellulose. Other sources include straw, corncobs, bagasse, and agricultural wastes.
Structure of Cellulose
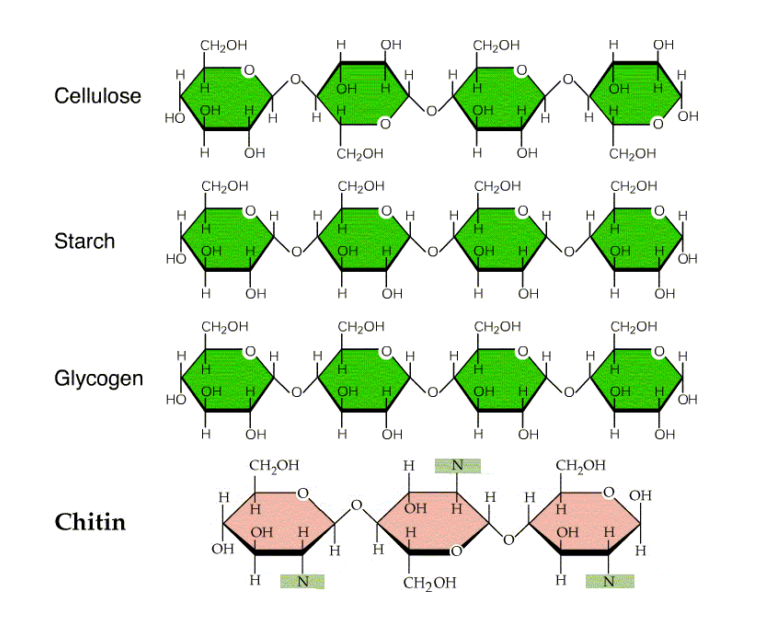
Cellulose is a linear homopolysaccharide structure consisting of D-glucose residues bonded by β(1→4)-O-glycosidic linkages. The structure of starch and cellulose differs fundamentally: cellulose is made up of β-glucose subunits, whereas starch contains α-glucose subunits.
Key structural features:
- Cellulose is unbranched and forms a straight-chain polymer of glucose
- The repeating unit in cellulose is cellobiose (two glucose molecules linked by β-1,4-glycosidic bonds)
- Long cellulose fibers are held together by intermolecular hydrogen bonds
- Hydrogen bonding between glucose units of adjacent chains creates strong, fibrous bundles
- This structure makes cellulose ideal for its structural function in plants
- Is cellulose a heteropolysaccharide? No, cellulose is a homopolysaccharide because it contains only glucose units.
- Is cellulose a homopolysaccharide? Yes, it consists of a single type of monosaccharide (glucose).
Properties of Cellulose
- Colorless amorphous solid with no melting point
- Decomposes under strong heating
- Insoluble in water and most organic solvents
- Dissolves in Schweitzer’s reagent (ammoniacal solution of cupric hydroxide)
- Humans lack enzymes for digesting cellulose, making it unsuitable for human nutrition but valuable as dietary fiber
B. Starch: The Storage Polysaccharide in Plants
Starch polysaccharide is the second most abundant carbohydrate in nature and the main contributor to carbohydrates in our diet. Is starch a polysaccharide? Yes, starch is a storage polysaccharide found only in plants, stored in seeds, roots, and fibers as a food reserve. The chief sources of starch are cereals, potatoes, corn, and rice.
Structure of Starch and Glycogen: Key Differences
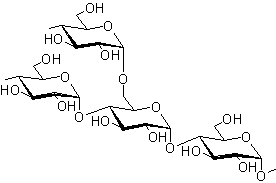
Starch is a polysaccharide composed of two structurally different components: Amylose (20%) and Amylopectin (80%). Both are composed of D-glucose units linked by α-glycosidic linkages.
Amylose Structure:
- Linear molecule formed by D-glucose units
- Linked by α(1→4)-glycosidic linkages between C-1 of one glucose unit and C-4 of the next
- Contains 60 to 300 D-glucose units
- Forms a helicoidal (helical) structure
- Water-soluble component of starch
Amylopectin Structure:
- Highly branched-chain structure
- Main chains of 25-30 D-glucose units joined by α(1→4)-glycosidic linkages
- Branch points connected by α(1→6)-glycosidic linkages
- Contains 300 to 6,000 D-glucose units
- Creates a tree-like branched structure
- The structure of starch, glycogen, and cellulose differs in branching frequency
Digestion products:
- Complete hydrolysis yields glucose
- Partial digestion produces maltose and isomaltose disaccharides
Is starch a heteropolysaccharide? No, starch is a homopolysaccharide because it contains only glucose monomers. Is starch a homopolysaccharide? Yes, both amylose and amylopectin are composed exclusively of glucose units.
C. Glycogen: Animal Starch
Is glycogen a polysaccharide? Yes, glycogen is a food storage polysaccharide often called “animal starch”. Glycogen polysaccharide serves as the primary glucose storage form in animals.
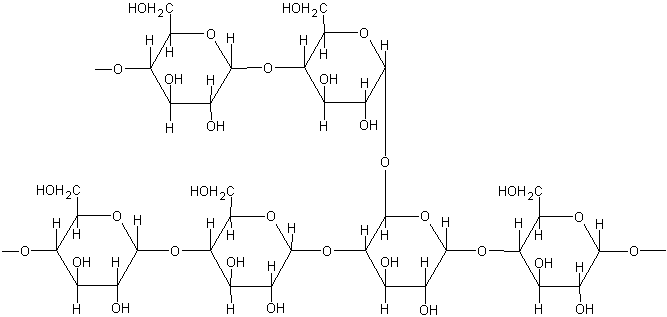
Glycogen Structure:
- Glycogen is polysaccharide stored mainly in liver and muscle tissues
- The structure of glycogen and starch is similar, but glycogen has more extensive branching
- Each branch chain contains 10-20 glucose units (compared to 25-30 in amylopectin)
- Branching occurs approximately every 8-12 glucose units via α(1→6)-glycosidic bonds
- Main chains linked by α(1→4)-glycosidic linkages
- Molecular weight: 1-2 × 10⁷
- More soluble in water than amylopectin due to extensive branching
Glycogen vs Starch Structure:
The difference between starch and glycogen structure lies primarily in branching frequency. Glycogen vs cellulose structure differs even more dramatically: glycogen contains α-glucose with extensive branching, while cellulose contains β-glucose in unbranched linear chains.
Biological Functions:
- Glycogen polysaccharide structure allows rapid glucose release when needed
- Liver glycogen: releases glucose into blood for systemic energy needs
- Muscle glycogen: provides local energy reserve for muscular contraction
- Hydrolysis yields maltose units
Is glycogen a homopolysaccharide? Yes, glycogen consists entirely of glucose monomers arranged in a highly branched structure.
The structure of glycogen is very similar to amylopectin but more branched, with one branch every 8 to 12 glucose units. Glycogen is the way in which glucose is stored in animals. Glycogen is stored mainly in the liver (to release glucose into the blood when necessary) and in muscles, where it is used as a reserve of energy for muscular contraction.
Starch Glycogen and Cellulose: Structural Comparison
Understanding the structure of starch glycogen and cellulose is essential for biochemistry students. Here’s a comprehensive comparison of these three important polysaccharides:
| Feature | Cellulose | Starch (Amylose/Amylopectin) | Glycogen |
|---|---|---|---|
| Monomer Type | β-glucose | α-glucose | α-glucose |
| Glycosidic Bonds | β(1→4) | α(1→4) and α(1→6) | α(1→4) and α(1→6) |
| Structure | Linear, unbranched | Amylose: linear; Amylopectin: branched | Highly branched |
| Branching Frequency | None | Every 25-30 units (amylopectin) | Every 8-12 units |
| Function | Structural support (plants) | Energy storage (plants) | Energy storage (animals) |
| Solubility | Insoluble in water | Partially soluble | Highly soluble |
| Location | Plant cell walls | Plant seeds, roots, tubers | Liver, muscles |
This comparison table clarifies the structural difference between starch and glycogen as well as the starch vs cellulose structure differences that determine their biological functions.
Heteropolysaccharides: Structure, Types and Functions
What is a heteropolysaccharide? Heteropolysaccharides contain two or more different types of monosaccharides. These complex polysaccharides provide extracellular support for organisms across all kingdoms—from the bacterial cell envelope to the matrix holding animal cells together.
Heteropolysaccharides and homopolysaccharides differ fundamentally: while homopolysaccharides contain only one monosaccharide type, heteropolysaccharides consist of diverse monosaccharide units, often including amino sugars and acidic sugars.
As a group, they perform diverse functions: structural, water metabolism regulation (as a reservoir of water), cellular cement, biological sieve, biological lubricant, and docking sites for growth factors, among other functions.
Classification of Heteropolysaccharides
Heteropolysaccharide classification includes several important groups based on structure and function:
Important heteropolysaccharides in the extracellular matrix:
- Hyaluronic acid (hyaluronate)
- Chondroitin sulfates (A, B, C)
- Dermatan sulfate
- Keratan sulfate
- Heparin
These heteropolysaccharides typically feature:
- Repeating disaccharide units of amino sugar and acidic sugar
- Sulfate groups linked to certain monosaccharides
- Association with proteins forming proteoglycans or glycosaminoglycans (also called mucopolysaccharides)
Heteropolysaccharide examples demonstrate diverse biological functions: structural support, water metabolism regulation, cellular cement, biological sieve, biological lubricant, and docking sites for growth factors.
a. Hyaluronic Acid (Hyaluronate)
Hyaluronic acid heteropolysaccharides are the most abundant mucopolysaccharides in higher animals, found in the vitreous body of the eye, umbilical cord, and synovial fluid of joints.
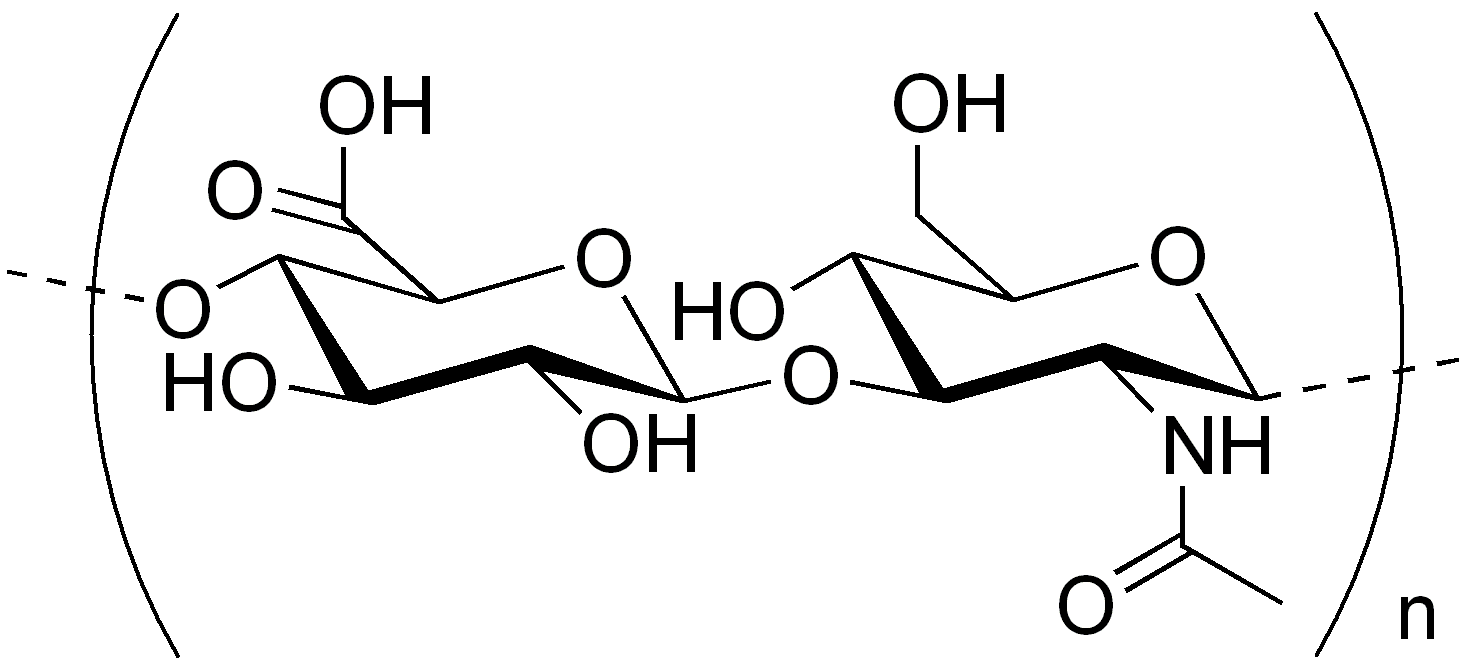
Structure and Properties:
- Straight-chain polymer alternating D-glucuronic acid and N-acetyl-D-glucosamine (NAG)
- Molecular weight: approximately 5,000,000
- Chain formed by β(1→3) and β(1→4) glycosidic linkages
- Acidic substance due to largely ionized carboxyl groups at cellular pH
Biological Functions:
- Biological lubricant in synovial fluid of joints (contains ~0.03% hyaluronic acid)
- Provides viscosity to synovial fluid
- Gives consistency to vitreous humor
- Contributes to tensile strength and elasticity of cartilage and tendons
b. Chondroitin Sulfates
Chondroitin sulfates are principal mucopolysaccharides in the ground substance of mammalian tissues and cartilage, occurring combined with proteins. Three types have been isolated:
Chondroitin Sulfate A:
- Chief component in cartilage, adult bone, and cornea
- Contains equimolar N-acetyl-galactosamine, D-glucuronic acid, and sulfuric acid
- Sulfate group at position 4 of galactosamine
Chondroitin Sulfate B (Dermatan Sulfate):
- Present in skin, heart valves, and tendons
- Contains L-iduronic acid instead of D-glucuronic acid
Chondroitin Sulfate C:
- Found in cartilage and tendons
- Similar to type A but with sulfate at position 6 of galactosamine
Chemical Structure:
- Repeating disaccharide unit: N-acetyl-D-galactosamine + uronic acid
- “Chondrosin” obtained by acid hydrolysis
- Molecular weight: ~43,300 (purified bovine cartilage)
c. Dermatan sulfate
Dermatan sulfate (formerly chondroitin sulfate B) is found mainly in skin, vessels, hearts, and lungs. It may relate to coagulation and vascular diseases.
Structure:
- Similar to chondroitin sulfate A except D-glucuronic acid is replaced by L-iduronic acid
- The two uronic acids differ in configuration only at C5
- Involves α(1→3) and β(1→4) linkages
- Dermatan sulfate is a heteropolysaccharide with alternating sugar units
d. Keratan sulfate
Structure and Composition:
- Differs from other mucopolysaccharides: uronic acid component replaced by D-galactose
- Contains N-acetyl-D-glucosamine esterified by sulfate at carbon 6
- Alternating linkages: β(1→4) and β(1→3)
- Linkage between repeating disaccharide units: β(1→3) (not β-1→4)
Location:
- Cornea, cartilage, bone
- Nails, hair, and various other structures
e. Heparin
Heparin is a blood anticoagulant present in the liver (original isolation source), lung, thymus, spleen, and blood.
Chemical Structure:
- Polymer of D-glucuronic acid and D-glucosamine
- Molecular weight: 17,000-20,000
- Strongly acidic due to sulfuric acid groups
- Readily forms salts (barium salt used in isolation)
Biological Function:
- Potent natural anticoagulant produced in mast cells
- Causes antithrombin to bind to thrombin
- Inhibits blood coagulation effectively
- Therapeutic applications in preventing blood clots
Final words about Polysaccharides
Polysaccharide types serve diverse and essential functions in living organisms. Structural polysaccharides like cellulose provide mechanical support, while storage polysaccharides such as starch and glycogen store energy.
- Glycosaminoglycans synthesis and degradation: Glycosaminoglycans are synthesized in the endoplasmic reticulum (ER) and Golgi apparatus. Lysosomal hydrolases break them down through specific enzymatic pathways.
- Clinical Significance—Mucopolysaccharidoses: Mucopolysaccharidosis results from a deficiency of one or more hydrolase enzymes needed to break down glycosaminoglycans.
In these hereditary disorders, glycosaminoglycans accumulate in tissues, causing:
- Skeletal deformities
- Extracellular matrix abnormalities
- Mental retardation
- Organ dysfunction
Examples of heteropolysaccharide storage diseases include:
- Hunter Syndrome: X-linked disorder affecting heparan sulfate and dermatan sulfate degradation
- Hurler Syndrome: Autosomal recessive disorder with similar biochemical defects
- Both diseases demonstrate the critical importance of proper glycosaminoglycan metabolism
What are polysaccharides? They are essential biological polymers that serve structural, storage, and signaling functions. Polysaccharide examples range from the cellulose in plant cell walls to the glycogen stored in your liver, demonstrating the remarkable diversity of these carbohydrate polymers.
Discover more from Biochemistry Den
Subscribe to get the latest posts sent to your email.





