The Krebs cycle, or the citric acid or TCA cycle, is a fundamental metabolic pathway that plays a critical role in cellular respiration. It occurs in the mitochondria of eukaryotic cells and involves the conversion of acetyl-CoA into ATP, the primary energy currency of the cell.
The Krebs cycle is named after Sir Hans Adolf Krebs, a German-born biochemist who described the pathway in 1937. The cycle is also called the citric acid cycle because citric acid is one of the intermediate molecules in the pathway. The term “TCA cycle” stands for the “tricarboxylic acid cycle,” which refers to the three-carbon molecules involved in the cycle.

In the following sections, we will explore the steps of the Krebs cycle, the enzymes involved, the significance of the cycle, and its regulation. By understanding the Krebs cycle, we can gain insight into the fundamental processes that sustain life and how they are regulated in living organisms.
What is Krebs Cycle?
The Krebs cycle, also known as the citric acid cycle or TCA cycle, is a fundamental metabolic pathway in living organisms that generates energy by breaking down food molecules.
It was first discovered by Sir Hans Adolf Krebs, a British biochemist, and is essential to the function of all aerobic organisms, including humans.
The Krebs cycle is essential for the breakdown of carbohydrates, fats, and proteins and for producing energy in the form of ATP. It is a complex and tightly regulated pathway that involves a series of enzymatic reactions that occur in a specific sequence. The cycle is a crucial part of cellular respiration, the process by which cells convert nutrients into energy to carry out their functions.
- Carbohydrates Classifications
- Glycolysis – Glucose Catabolic Pathway
- Carbohydrate Metabolism: Glycogenesis
In this article, we will discuss the steps involved in the Krebs cycle and how it helps in generating energy in our cells. Additionally, we will provide a diagram of the Krebs cycle for better understanding.
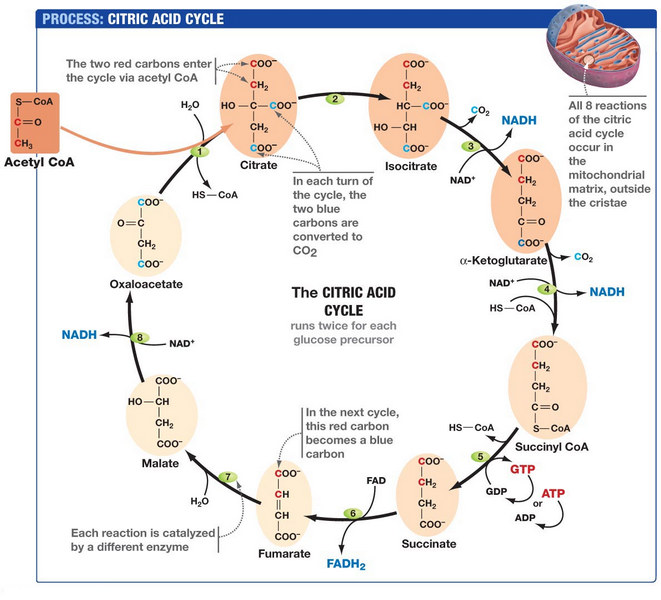
Steps of the Krebs Cycle
The Krebs cycle involves a series of chemical reactions that take place in the mitochondria of eukaryotic cells. It begins with the breakdown of glucose, a simple sugar that is an essential source of energy for the body. Here are the steps involved in the Krebs cycle:
The Krebs cycle, also known as the citric acid cycle or TCA cycle, is a complex metabolic pathway in eukaryotic cells’ mitochondria. The cycle involves nine enzymatic reactions that occur in a specific order to generate energy in the form of ATP. Here are the steps of the Krebs cycle with proper side headings and chemical equations:
Step 1: Acetyl-CoA Formation
The Krebs cycle begins with the Formation of acetyl-CoA from pyruvate, which is produced by glycolysis. The reaction is catalyzed by the enzyme pyruvate dehydrogenase and is as follows:
Pyruvate + CoA + NAD+ → Acetyl-CoA + NADH + CO2
Step 2: Citrate Formation
Acetyl-CoA then combines with oxaloacetate to form citrate, a six-carbon molecule, in a reaction catalyzed by citrate synthase. The chemical equation is:
Acetyl-CoA + oxaloacetate + H2O → citrate + CoA
Step 3: Isocitrate Formation
Citrate is then isomerized to isocitrate by the enzyme aconitase. The chemical equation is:
citrate ↔ isocitrate
Step 4: α-Ketoglutarate Formation
Isocitrate is oxidized to α-ketoglutarate in a reaction catalyzed by isocitrate dehydrogenase. The chemical equation is:
isocitrate + NAD+ + H2O → α-ketoglutarate + NADH + CO2
Step 5: Succinyl-CoA Formation
α-Ketoglutarate is oxidized to succinyl-CoA in a reaction catalyzed by α-ketoglutarate dehydrogenase. The chemical equation is:
α-ketoglutarate + CoA + NAD+ → succinyl-CoA + NADH + CO2
Step 6: Succinate Formation
Succinyl-CoA is converted to succinate in a reaction catalyzed by succinyl-CoA synthetase, which produces ATP. The chemical equation is:
Succinyl-CoA + ADP + Pi → succinate + CoA + ATP
Step 7: Fumarate Formation
Succinate is then oxidized to fumarate in a reaction catalyzed by succinate dehydrogenase, which is also part of the electron transport chain. The chemical equation is:
Succinate + FAD → fumarate + FADH2
Step 8: Malate Formation
Fumarate is then hydrated to form malate in a reaction catalyzed by fumarase. The chemical equation is:
Fumarate + H2O → malate
Step 9: Oxaloacetate Formation
Malate is then oxidized to oxaloacetate in a reaction catalyzed by malate dehydrogenase, generating NADH. The chemical equation is:
Malate + NAD+ → oxaloacetate + NADH + H+
Overall, the Krebs cycle generates 1 ATP, 3 NADH, 1 FADH2, and 2 CO2 molecules per acetyl~CoA molecule that enters the cycle. These high-energy molecules are then used in the electron transport chain to generate ATP through oxidative phosphorylation.
Diagram of the Krebs Cycle:
The Krebs cycle is a highly regulated process, with each step being catalyzed by a specific enzyme. In addition to producing energy-rich molecules like NADH and FADH2, the cycle also helps to replenish oxaloacetate, a molecule that is used up in the initial step of the cycle.
Here is a diagram of the Krebs cycle that will help you understand the steps involved in the process:
Net ATP Calculation of Citric acid cycle
| Step number | Reactions | Coenzyme | ATP generated |
| Step 4 & 5 | Isocitrate -> α-ketoglutarate | NADH | 3 |
| Step 6 | α-ketoglutarate -> Succinyl~coA | NADH | 3 |
| Step 7 | Succinyl ~ coA -> Succinate | GTP or ATP | 1 |
| Step 8 | Succinate -> Fumarate | FADH2 | 2 |
| Step 10 | Malate -> Oxaloacetate | NADH | 3 |
Inhibitors of TCA cycle
- Aconitase is inhibited by Flouro Acetate. This is a non-competitive inhibition.
- α-ketoglutarate dehydrogenase is inhibited by Arsinite. This again is a non-competitive inhibition.
- Succinate dehydrogenase is inhibited by Malonate. This is a competitive inhibition.
Significance of the Krebs cycle
The Krebs cycle, also known as the citric acid cycle or TCA cycle, is a vital metabolic pathway that plays a crucial role in producing ATP, the primary energy source for cells. Here are some of the vital significance of the Krebs cycle:
a. Energy Production
The Krebs cycle produces energy-rich molecules like ATP, NADH, and FADH2, essential for cellular respiration. The electron transport chain uses these molecules to produce ATP, which is used for various cellular processes.
b. Metabolism of Carbohydrates
The Krebs cycle is involved in the metabolism of carbohydrates. Glucose is broken down into pyruvate, which is then converted into acetyl-CoA and fed into the Krebs cycle. The cycle helps in the complete oxidation of glucose, which results in the production of ATP.
c. Metabolism of Fats
The Krebs cycle is also involved in the metabolism of fats. Fatty acids are broken down into acetyl-CoA and then fed into the Krebs cycle for further oxidation. This process helps in the production of ATP.
d. Biosynthesis of Molecules
The Krebs cycle involves the biosynthesis of several molecules like amino acids, nucleotides, and heme. These molecules are essential for various cellular processes, including DNA and protein.
e. Regulation of pH
The Krebs cycle helps in the regulation of pH in the mitochondria. The cycle produces carbon dioxide, which can act as a buffer and maintain the pH of the mitochondria.
This is a fundamental metabolic pathway that plays a crucial role in energy generation and the metabolism of various biomolecules. Understanding the significance of the Krebs cycle is essential for gaining a deeper understanding of cellular metabolism and the functioning of living organisms.
Regulation of Krebs Cycle
The Krebs cycle, also known as the citric acid or TCA cycle, is a highly regulated metabolic pathway that plays a crucial role in cellular respiration. Here are some of the key ways in which the Krebs cycle is regulated:
a. Feedback Inhibition
The Krebs cycle is regulated by feedback inhibition, in which the end products of the cycle inhibit earlier enzymes in the pathway. For example, high levels of ATP can inhibit the enzyme isocitrate dehydrogenase, which is involved in the third step of the cycle.
b. Substrate Availability
The availability of substrates like acetyl-CoA and oxaloacetate can also regulate the Krebs cycle. The cycle may slow down or even stop if there is a shortage of these substrates.
c. Hormonal Regulation
Hormones like insulin and glucagon can also regulate the Krebs cycle. Insulin can increase the activity of enzymes like pyruvate dehydrogenase and citrate synthase, while glucagon can decrease their activity.
d. Allosteric Regulation
Allosteric Regulation occurs when a molecule binds to an enzyme and changes its shape, altering its activity. For example, NADH can bind to the enzyme isocitrate dehydrogenase and inhibit its activity.
e. Redox State
The redox state of the cell can also regulate the Krebs cycle. High levels of NADH and ATP can inhibit the cycle, while low levels of these molecules can stimulate it.
f. Genetic Regulation
The Krebs cycle is regulated by the expression of genes that encode the enzymes involved in the pathway. Changes in gene expression can alter the activity of the Krebs cycle and affect cellular metabolism.
The Regulation of the Krebs cycle is a complex process involving various mechanisms. By understanding these mechanisms, researchers can gain a deeper insight into the functioning of living organisms and the fundamental processes that sustain life.
Frequently Answered Questions (FAQs)
What is the Krebs cycle?
The Krebs cycle is a metabolic pathway that occurs in the mitochondria of eukaryotic cells. It involves a series of enzyme-catalyzed reactions that break down food molecules to generate energy in the form of ATP.
What is the significance of the Krebs cycle?
The Krebs cycle is an essential part of cellular respiration and plays a critical role in generating energy for our cells. It breaks down food molecules to release energy in the form of ATP, which is used for various cellular processes.
What happens if the Krebs cycle is disrupted?
Disruptions to the Krebs cycle can lead to various metabolic disorders, such as lactic acidosis, which is caused by a buildup of lactic acid in the body. Additionally, some genetic mutations can affect the enzymes involved in the Krebs cycle, leading to a range of health problems.
What are the other names for the Krebs cycle?
The Krebs cycle is also known as the citric acid cycle or the TCA cycle, named after the scientist who discovered it, Sir Hans Adolf Krebs.
Conclusion
The Krebs cycle is a fundamental metabolic pathway that is crucial for generating energy in our cells. It involves a series of enzyme-catalyzed reactions that break down food molecules to produce ATP.
The diagram of the Krebs cycle provided in this article can help you understand the process better. Understanding the steps of the Krebs cycle and its significance can help us appreciate the complexity of the processes that occur in our cells.
Discover more from Biochemistry Den
Subscribe to get the latest posts sent to your email.