Carbohydrates are a class of organic molecules with the general chemical formula Cn(H₂O)n. They are composed of carbon, hydrogen, and oxygen and perform many important roles in living organisms.
They serve as fuel for cellular respiration, act as the main source of energy for most organisms, store energy in the form of chemical bonds, and form structural components of some cells. In this blog, we will discuss the color reactions of carbohydrates.
Carbohydrates are literally “carbon hydrates” of monomers. A number of simple qualitative tests can be used to characterize sugars. These tests use specific reagents that yield a color change after reacting with particular functional groups of the compounds being tested.
These reactions can detect the presence or absence of carbohydrates in test solutions and range in specificity from very general to highly specific. In this experiment, the Molisch test, iodine test, Benedict’s test, Barfoed’s test, Seliwanoff’s test, and Bial’s test are performed to detect specific carbonyl groups in carbohydrates. Several qualitative tests have been devised to detect members of this biologically significant class of compounds.
You are given solutions containing fructose, glucose, lactose, galactose, ribose, ribulose, sucrose, and starch. Using the principles described, you can devise a scheme to systematically identify these compounds.

You are given solutions containing: fructose, glucose, lactose, galactose, ribose, ribulose, sucrose, and starch. Devise a scheme by which you can systematically identify these compounds.
Introduction
Simple sugars, starches, and cellulose are organic compounds that have the approximate formula C(H₂O)n, which explains the name “carbohydrates” or “hydrates of carbon” that is usually applied to this group.
They are not true hydrates of carbon but polyhydroxy alcohol compounds that contain an aldehyde or ketone functional group. These functional groups give carbohydrates many of their characteristic chemical properties. Several qualitative tests have been developed to detect members of this biologically significant class of compounds.
These tests use reagents that produce a color change after reacting with specific functional groups in the molecules being tested. These reactions can detect the presence or absence of carbohydrates in test solutions and range from very general to highly specific.
The Chemical Test and Procedure
| Test | Procedure |
| Iodine Test | + 10 drops iodine reagent, observe |
| Benedict’s | + 10 drops Benedict’s reagent, water bath (100 degrees Celsius) |
| Barfoed’s | + 10 drops Barfoed’s reagent, water bath (100 degrees Celsius) |
| Seliwanoff’s | + 10 drops Seliwanoff’s reagent, water bath (100 degrees Celsius) |
| Bial’s | + 10 drops of Bial’s reagent, water bath (100 degrees Celsius) |
Carbohydrates are found in fruits, vegetables, grains, beans, and dairy foods, and they are important because they provide energy for our bodies. The color reactions table below summarizes how certain carbohydrates react with different reagents under defined conditions.
For example, lactose is milk sugar. It is found naturally in milk. If lactose was to be dissolved in a water solution, it would turn into a milky liquid. This reaction occurs when milk is heated and left to stand for a prolonged time at room temperature. Lactose is also called milk sugar.
What is the basic procedure for the color reaction of carbohydrates?
Perform the following qualitative tests on 0.2 M solutions (unless otherwise stated) of starch, sucrose, glucose, lactose, galactose, ribose, and ribulose. Use the scheme you devised in the pre‑lab section to identify an unknown solution.
The unknown will be one of the above solutions or a mixture of two of them.
Test 1: Molisch’s Test for Carbohydrates
The Molisch test is a general test for the presence of carbohydrates. Molisch’s reagent is a solution of α‑naphthol in 95% ethanol. This test is useful for identifying any compound that can be dehydrated to furfural or hydroxymethylfurfural in the presence of concentrated H₂SO₄..
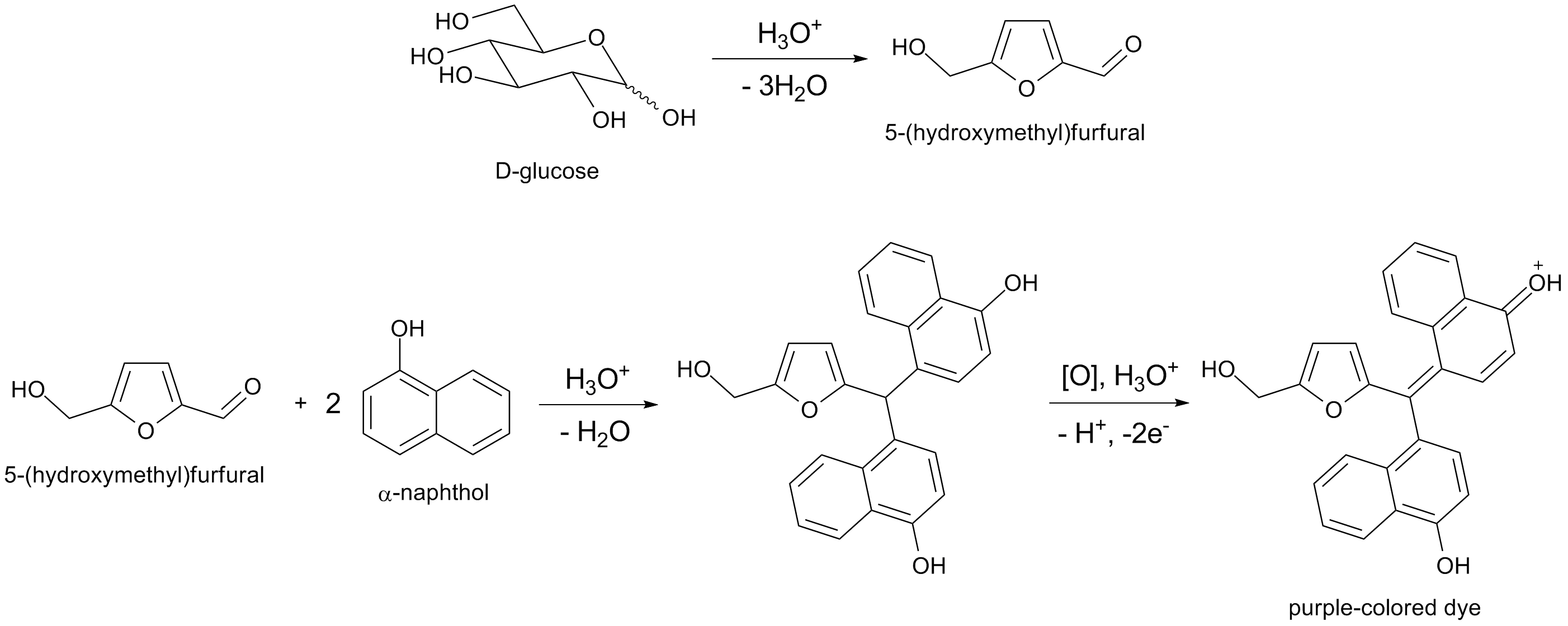
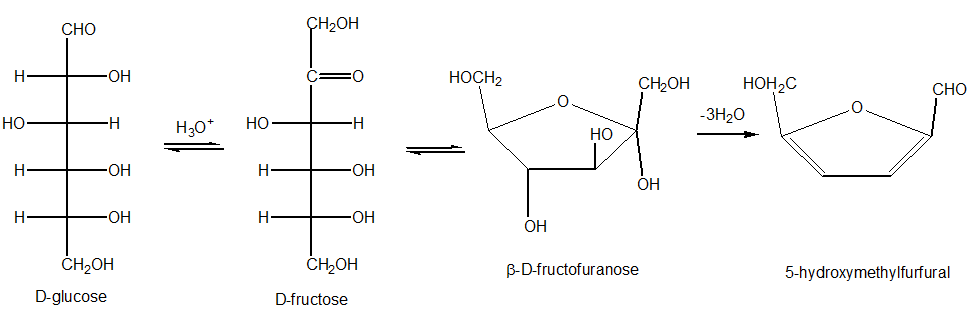
Furfural is formed by dehydration of pentoses and pentosans, whereas hydroxymethylfurfural is formed from hexoses and hexosans. Oligosaccharides and polysaccharides are hydrolyzed by the acid to yield their monomeric units.
The α‑naphthol reacts with these cyclic aldehydes to form purple condensation products. Although this test can detect compounds other than carbohydrates (for example, glycoproteins), a negative result strongly suggests the absence of carbohydrates.
Method: Add 2 drops of Molisch’s reagent to 2 mL of the sugar solution and mix thoroughly. Incline the tube and gently pour 5 mL of concentrated H₂SO₄ down the side of the tube so that it forms a separate layer. A purple color at the interface of the sugar solution and acid indicates a positive test. Ignore any green color that may appear.
Test 2: Benedict’s Test for Reducing Sugars
Alkaline solutions of copper are reduced by sugars that have a free aldehyde or ketone group, leading to the formation of colored cuprous oxide. Benedict’s solution contains copper sulfate, sodium carbonate, and sodium citrate at approximately pH 10.5. The citrate forms soluble complexes with Cu²⁺, preventing precipitation of CuCO₃ in the alkaline solution.
Method: Add 1 mL of the solution to be tested to 5 mL of Benedict’s solution and shake the tube. Place the tube in a boiling water bath and heat for about 3 minutes. Remove the tube from the bath and allow it to cool. Formation of a green, yellow, or red precipitate indicates a positive test for reducing sugars.
Test 3: Barfoed’s Test for Monosaccharides
This reaction detects reducing monosaccharides in the presence of disaccharides. Barfoed’s reagent contains copper acetate in dilute acetic acid at about pH 4.6 and uses copper ions to detect reducing sugars in an acidic solution. The color changes observed are similar to those seen in Benedict’s test.
Method: Add 1 mL of the solution to be tested to 3 mL of freshly prepared Barfoed’s reagent. Place the tube in a boiling water bath and heat for 3 minutes, then remove and allow it to cool.
Formation of a green, yellow, or red precipitate within 3 minutes indicates a positive test for reducing monosaccharides. Do not heat the tubes longer than 3 minutes; if disaccharides are heated for too long, they may also give a positive result.
Test 4: Lasker and Enkelwitz Test for Ketoses
The Lasker and Enkelwitz test uses Benedict’s solution, but the reaction is carried out at a much lower temperature. The color changes observed are the same as in the standard Benedict’s test. Dilute sugar solutions (about 0.02 M) should be used.
Method: Add 1 mL of the solution to be tested to 5 mL of Benedict’s solution in a test tube and mix well. Heat the tube in a 55°C water bath for 10–20 minutes. Ketopentoses give a positive reaction within about 10 minutes, while ketohexoses require about 20 minutes. Aldoses do not give a positive reaction under these conditions.
Test 5: Bial’s Test for Pentoses
Bial’s reagent contains orcinol, HCl, and FeCl₃.. Orcinol forms colored condensation products with furfural generated by the dehydration of pentoses and pentosans. Dilute sugar solutions (about 0.02 M) are required for this test.

Method: Add 2 mL of the solution to be tested to 5 mL of Bial’s reagent. Gently heat the tube to boiling, then allow it to cool. Formation of a green solution or precipitate indicates a positive reaction for pentoses.
Test 6: Mucic Acid Test for Galactose
Oxidation of most monosaccharides by nitric acid produces soluble dicarboxylic acids. However, oxidation of galactose yields an insoluble dicarboxylic acid called mucic acid. Lactose also yields mucic acid because nitric acid hydrolyzes the glycosidic bond between glucose and galactose, releasing free galactose.
Method: Add 1 mL of concentrated nitric acid to 5 mL of the solution to be tested and mix well. Heat the mixture in a boiling water bath until the volume is reduced to about 1 mL. Remove the tube from the bath and allow it to stand at room temperature overnight. The appearance of insoluble crystals at the bottom of the tube indicates the presence of mucic acid.
Test 7: Iodine Test for Starch and Glycogen
Lugol’s iodine reagent is useful for distinguishing starch and glycogen from other polysaccharides. Lugol’s iodine produces a blue‑black color in the presence of starch.
Glycogen reacts with Lugol’s reagent to produce a brown‑blue color. Other polysaccharides and monosaccharides cause no significant color change, and the solution remains the characteristic brown‑yellow of the reagent.
It is thought that starch and glycogen form helical coils into which iodine molecules can fit, forming starch‑iodine or glycogen‑iodine complexes.
Starch (amylose and amylopectin) has fewer branches than glycogen, so its helices are longer and can bind more iodine atoms. As a result, the color produced by a starch‑iodine complex is more intense than that produced by a glycogen‑iodine complex.
Method: Add 2–3 drops of Lugol’s iodine solution to 5 mL of the solution to be tested. Starch gives a blue‑black color. A positive test for glycogen is a brown‑blue color. A negative test is indicated by the unchanged brown‑yellow color of the reagent.
A negative test is the brown-yellow color of the test reagent.
- What are Polysaccharides and How it was Classified?
- What are the Classifications of Carbohydrates? Explain with Examples
- Carbohydrates are the Staff of Life. Why?
- What are Monosaccharides? How to Classify?
Positive Reactions for Carbohydrates Test
Molisch | Deep purple / purple ring at interface |
Iodine | Black / blue‑black |
Benedict | Brick‑red precipitate |
| Barfoed | Brick‑red precipitate |
| Seliwanoff | Deep cherry red |
| Bial’s test | Yellow‑orange (often reported as blue‑green) |
The reaction of Carbohydrate Samples
Samples | Molisch | Iodine | Benedict | Barfoed | Seliwanoff | Bial |
| Glucose | + | – | + | ++ | – | – |
| Fructose | ++ | – | + | ++ | ++ | ++ |
| Ribose | – | – | – | – | – | – |
| Lactose | + | – | + | – | – | – |
| Sucrose | ++ | – | – | – | – | ++ |
| Starch | ++ | ++ | – | – | – | – |
| Unknown 1 | + | – | ++ | ++ | ++ | ++ |
| Unknown 2 | + | – | + | – | – | – |
| Identity of Unknown 1: | Fructose | |||||
| Identity of Unknown 2: | Lactose |
Qualitative Analysis protocols
Quantitative Analysis protocols
- Assay of Acid Phosphatase enzyme activity from Potatoes
- Assay of Urease Enzyme Activity (Enzymology Practical Protocol)
- Effect of Temperature on Amylase activity (Enzymology Protocol)
- Assay of Salivary Amylase enzyme activity
- Determination of Carbohydrate by Anthrone Method
Legend: (++) = fast reaction/ (+ )=slow reaction/ (-) is no reaction
Frequently Asked Questions (FAQs)
What color change indicates the presence of glucose?
In Benedict’s test, a positive glucose result changes from blue to green, yellow, or brick‑red, depending on the concentration.
What reagent is used to test for carbohydrates?
Benedict’s reagent is commonly used to test for many simple carbohydrates that act as reducing sugars. When it reacts with reducing sugars, it changes from blue to green, yellow, or orange‑red.
What color is a positive test for starch?
In the iodine test, adding yellow‑brown iodine solution to starch produces a blue‑black color, indicating a positive result for starch.
What is the general test for carbohydrates?
The Molisch test is the general test for carbohydrates, as it detects all compounds that can be dehydrated to furfural or hydroxymethylfurfural. Benedict’s test is a general test for reducing sugars, which are simple carbohydrates with free aldehyde or ketone groups.
What color does iodine turn in the presence of glucose?
Iodine does not give a characteristic color with glucose; it remains brown‑yellow. In contrast, iodine produces a blue‑black color with starch, allowing you to distinguish starch from glucose and other simple sugars.
What reagent is used to test for reducing sugars?
Benedict’s solution is widely used to test for reducing sugars.
What color does glucose turn with an iodine test?
Glucose does not produce a significant color change with iodine, so the solution remains brown‑yellow. The blue‑black color appears only when iodine reacts with starch.
Why does iodine not react with glucose?
Iodine reacts with the helical structure of amylose in starch, forming a starch–iodine complex that appears blue‑black. Glucose lacks this helical polymer structure, so iodine does not form a similar complex and no characteristic color appears.
Why do we add hydrochloric acid when testing for non-reducing sugars?
Hydrochloric acid hydrolyzes non‑reducing sugars (such as sucrose) into their monosaccharide components by breaking the glycosidic bonds. These monosaccharides are then reducing sugars and can be detected by Benedict’s reagent.
Why does glycogen give a red color with iodine?
In the iodine test, starch forms a blue‑black complex with iodine, while glycogen forms a reddish‑brown complex. The difference arises from the more highly branched, shorter helical structure of glycogen compared with starch.
Why does Benedict’s test require heat?
Heating provides the activation energy needed for the redox reaction in which reducing sugars reduce Cu²⁺ to Cu⁺, forming a colored precipitate of cuprous oxide.
Which sugar gives a positive color reaction with the mucic acid test?
Galactose gives a positive mucic acid test, forming insoluble mucic acid crystals. Lactose also gives a positive result because it is hydrolyzed to galactose and glucose.
Which color appears as Benedict’s reagent reacts?
Benedict’s reagent changes from blue to green, yellow, orange, or brick‑red, depending on the amount of reducing sugar present.
Cellulose gives which color with iodine?
Cellulose generally does not form a characteristic colored complex with iodine, so the solution remains the brown‑yellow color of the iodine reagent.
Which reagent is used to detect the presence of starch?
Iodine solution (Lugol’s iodine) is used to detect the presence of starch, producing a blue‑black color when starch is present.
Get this protocol in PDF format. Just download this “Color Reactions of Carbohydrates” file and make a print and distribute to the students. It helps you to protect your students from spelling mistakes and volumetric errors. All the best
Discover more from Biochemistry Den
Subscribe to get the latest posts sent to your email.