Ion exchange chromatography is a liquid chromatography technique used to separate ions and polar molecules based on their net charge. A sample containing charged molecules is passed through a column packed with an ion exchange resin, which carries fixed positive or negative charges.
Molecules with the opposite charge are attracted to the resin and temporarily retained, while others pass through the column.

By changing the pH or salt concentration of the mobile phase, these interactions can be weakened, and the bound molecules are eluted in an orderly way.
Because of its high resolution, flexibility, and scalability, ion exchange chromatography is a central method in biomolecule purification and analytical separation.
Basic Principle of Ion Exchange Chromatography
The principle of ion exchange chromatography is the reversible exchange of ions between charged groups on a stationary phase and oppositely charged ions in the mobile phase.
The stationary phase is an ion exchange resin with covalently attached ionic groups, while the mobile phase is an aqueous buffer containing counter‑ions and the sample molecules.
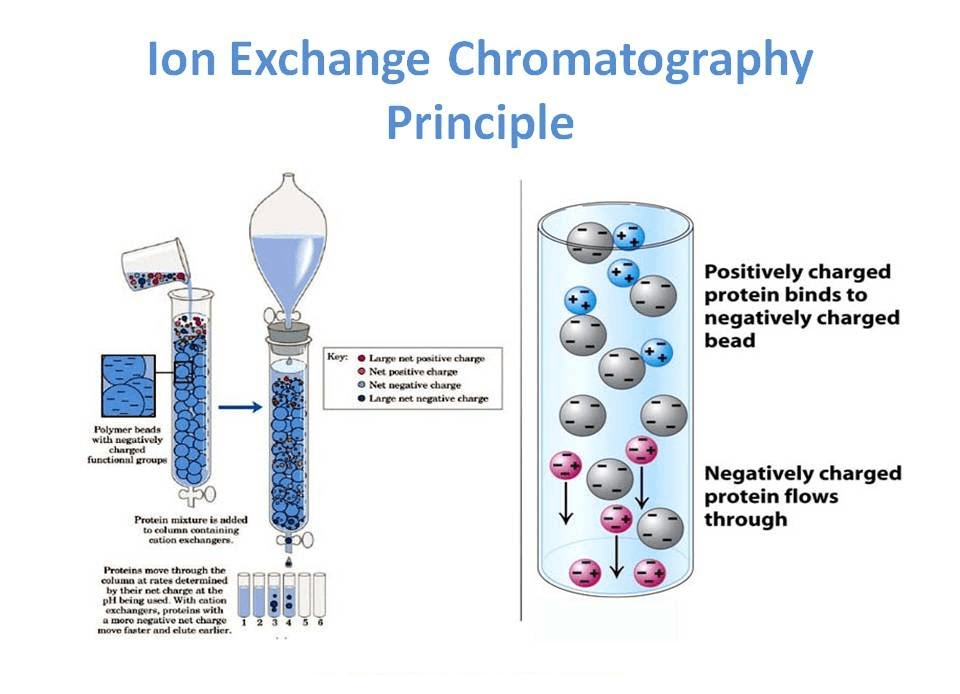
When the sample enters the column, molecules with a net charge complementary to the resin bind through electrostatic attraction.
The strength of binding depends on the sign and magnitude of the molecule’s net charge, as well as the pH and ionic strength of the buffer.
During elution, increasing salt concentration or adjusting pH introduces competing ions that displace the bound molecules from the resin, allowing them to elute at different volumes according to their charge properties.
Types of Ion Exchange Chromatography
Ion exchange chromatography is broadly divided into two main types, depending on the charge of the stationary phase.
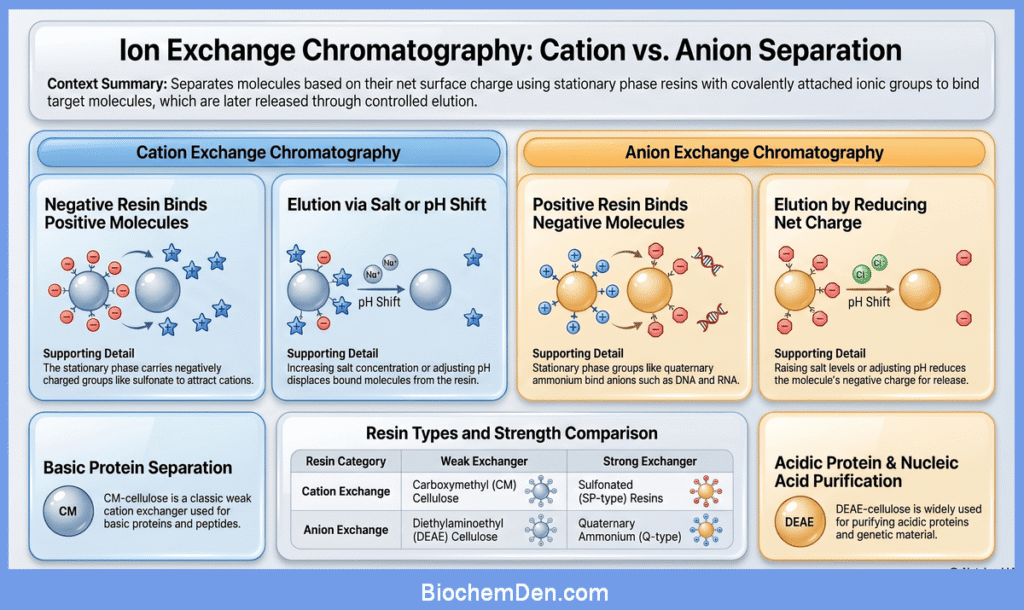
a. Cation Exchange Chromatography
In cation exchange chromatography, the resin carries negatively charged functional groups such as sulfonate or carboxymethyl groups.
These resins bind positively charged molecules (cations), including many proteins that are positively charged at the selected pH.
During elution, cations are displaced from the resin by increasing salt concentration or by shifting the pH so that the net charge on the molecules decreases.
Carboxymethyl cellulose (CM‑cellulose) is a classic weak cation exchanger used for separating basic proteins and peptides.
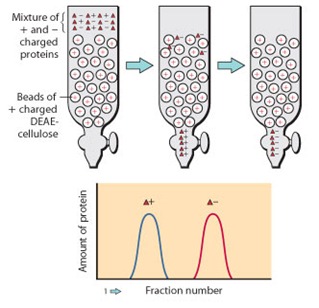
b. Anion Exchange Chromatography
In anion exchange chromatography, the resin carries positively charged groups, such as quaternary ammonium or diethylaminoethyl (DEAE) groups.
These resins bind negatively charged molecules (anions), including many acidic proteins and nucleic acids.
Elution is accomplished by raising the salt concentration or adjusting pH so that the net negative charge of the bound molecules is reduced and they are displaced from the resin.
DEAE‑cellulose is a widely used weak anion exchanger for the purification of DNA, RNA, and many acidic proteins.
Ion Exchange Resins and Stationary Phase
Ion exchange resins are insoluble, porous beads made from cross‑linked polymers such as polystyrene or cellulose, with fixed ionic groups covalently attached to their surface.
These ionic groups determine whether the resin behaves as a cation or anion exchanger and define its capacity and selectivity.
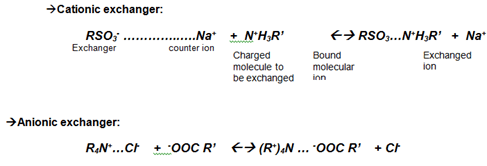
Common examples include:
- Cation exchangers
- Carboxymethyl (CM) cellulose: a weak cation exchanger with carboxymethyl groups.
- Sulfonated resins (for example, SP‑type resins): strong cation exchangers with sulfonate groups.
- Anion exchangers
- Diethylaminoethyl (DEAE) cellulose: a weak anion exchanger with DEAE groups.
- Quaternary ammonium resins (for example, Q‑type resins): strong anion exchangers.
The choice of resin depends on the pH range, the charge of the target molecule, and the required binding strength and resolution.
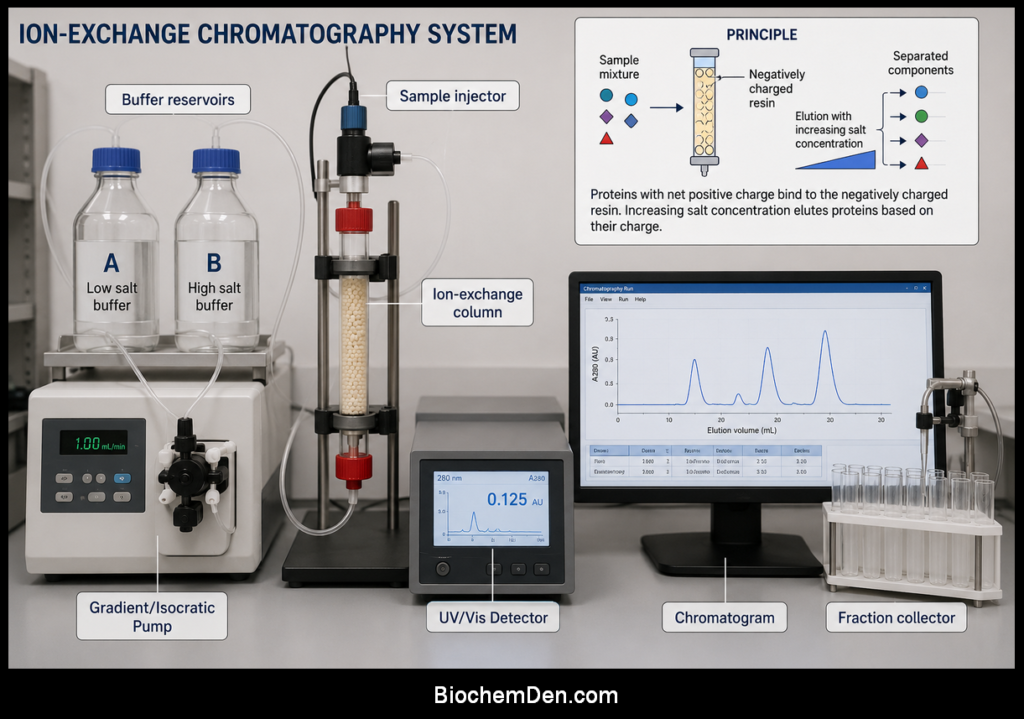
Components and Instrumentation of Ion Exchange Chromatography
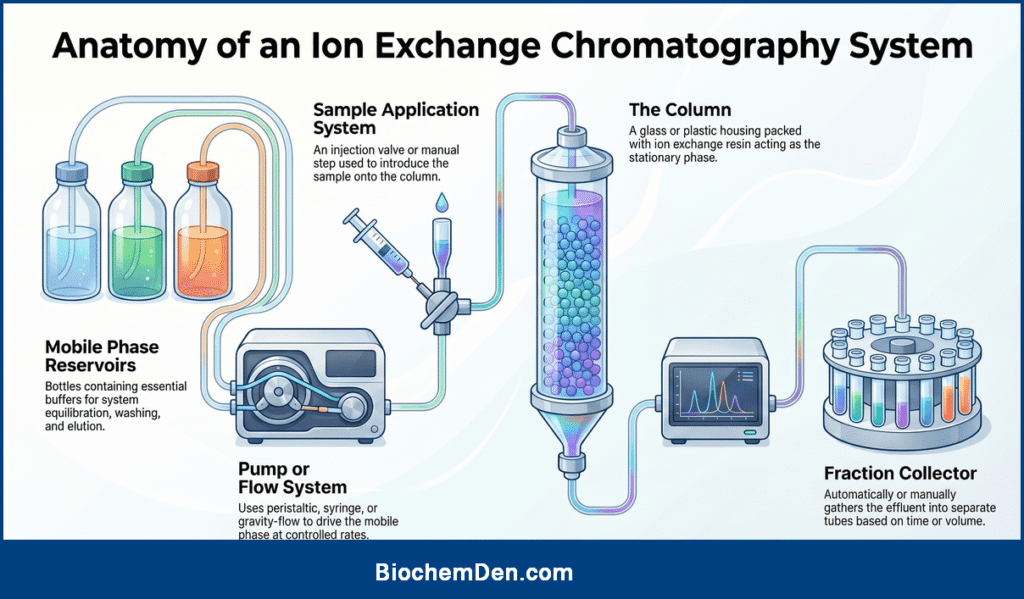
A typical ion exchange chromatography system consists of the following components:
- Column: A glass or plastic column packed with ion exchange resin, providing the stationary phase.
- Mobile phase reservoirs: Bottles containing buffers for equilibration, washing, and elution.
- Pump or flow system: A peristaltic pump, syringe pump, or gravity‑flow arrangement to drive the mobile phase through the column at controlled flow rates.
- Sample application system: An injection valve or manual loading step for introducing the sample onto the column.
- Detector: Typically a UV–Vis or conductivity detector to monitor the presence of eluting components.
- Fraction collector: An automated or manual collector that gathers column effluent into separate tubes according to time or volume.
In modern systems, ion exchange chromatography can also be integrated into HPLC setups with high‑pressure pumps, gradient mixers, and computer‑controlled data acquisition.
Ion-Exchange Chromatography (Step‑by‑Step Procedure)
Ion‑exchange chromatography is usually carried out in a series of standardized steps. In practice, protocols from different companies and laboratories all follow the same core sequence: buffer preparation, column equilibration, sample loading, washing, elution, and column regeneration.
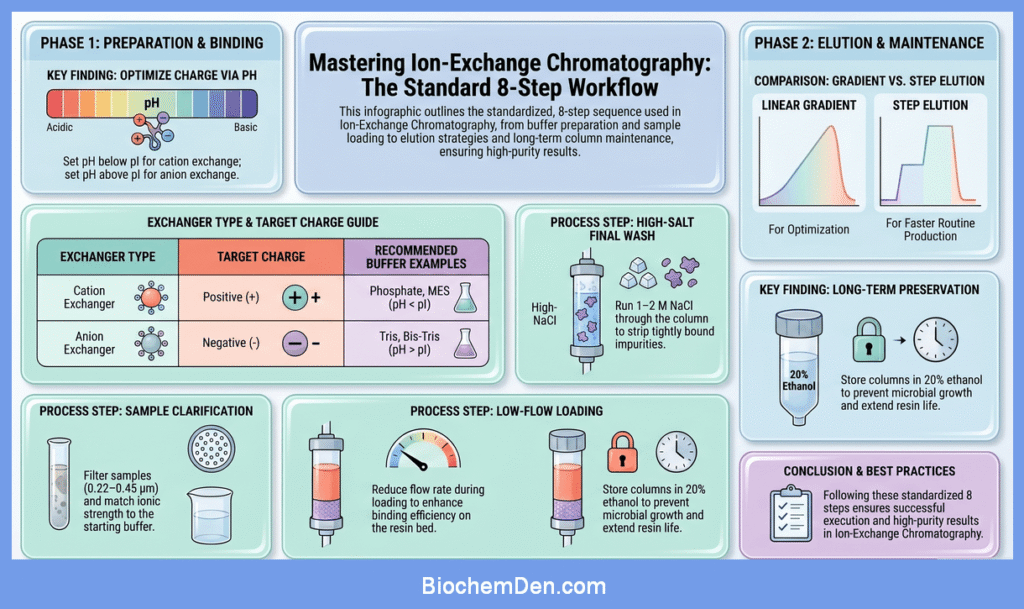
1. Buffer Preparation
Before starting, prepare all buffers carefully, because pH and ionic strength strongly control binding and elution.
- Choose a buffer system compatible with your target molecule and the ion‑exchange resin (for example, phosphate or MES for cation exchangers; Tris or bis‑Tris for anion exchangers).
- Adjust the pH so that the target molecule has an appropriate net charge:
- For cation exchange chromatography, set the pH below the isoelectric point (pI) of the protein so it is positively charged.
- For anion exchange chromatography, set the pH above the pI so the molecule is negatively charged.
- Keep the starting buffer at relatively low ionic strength (often 0–50 mM salt) to promote binding.
- After adding salts, re‑check and readjust the pH, because salt additions can shift pH.
Typically, you will prepare at least two buffers:
- Buffer A: low‑salt starting and wash buffer.
- Buffer B: high‑salt buffer for gradient or step elution.
| Buffers | PH range |
| Ammonium acetate | 4 to 6 |
| Ammonium formate | 3 to 5 |
| Pyridinium formate | 3 to 6 |
| Pyridinium acetate | 4 to 6 |
| Ammonium carbonate | 8 to 10 |
2. Column Preparation and Equilibration
If you are packing your own column, follow the resin manufacturer’s instructions for slurry preparation, degassing, and packing. Prepacked columns can be connected directly after a brief rinse.
- Connect the column to the chromatography system or set it up for gravity flow.
- Flush the column with water or appropriate storage solution to remove preservatives.
- Equilibrate the column with about 3–10 column volumes (CV) of Buffer A until the pH and conductivity at the outlet are stable and match the incoming buffer.
At the end of this step, all functional groups on the resin should be in the correct ionic form, and the column should be ready to bind the target molecules.
Preparation of the exchange medium
There are three steps of absolute importance:
1. Swelling of medium: (Pre-cycling)
Swelling makes the functional groups exposed for ion exchange.
- The swelling of anion exchangers is usually carried out by treating it. first with an acid (0.5N HCl) and then with base (0.5N NaOH).
- Exactly the reverse is the case with cationic exchangers. The matrix can be treated with EDTA for impurity eliminations.
2. Removal of very small particles
These fines will decrease the flow rate and the unsatisfactory reaction. To remove fines, the exchanger is repeatedly suspended in a large volume of water, and after the larger polymers have settled down, the slow-sedimenting materials are decanted.
3. Equilibration with counterions
This is accomplished by washing the exchanger with different reagents depending upon the desired counterion to be introduced.
- NaOH –> counter ion to be introduced is “Na⁺”
- HCl –> counter ion to be introduced is “H⁺”
- NaNO₃ –> counter ion to be introduced is “NO₃”
3. Sample Preparation
Preparing the sample correctly increases binding efficiency and resolution.
- Clarify the sample by centrifugation and/or filtration (for example, 0.22–0.45 µm) to remove particulates that could clog the column.
- Adjust the sample buffer so that pH and ionic strength closely match Buffer A used for column equilibration.
- If the original sample is in a high‑salt buffer or different pH, use dialysis or desalting columns to exchange it into the ion‑exchange starting buffer.
- Keep the sample at a temperature that preserves stability (often 2–8 °C for proteins) until loading.
The sample is now in conditions that allow the target molecules to bind strongly to the resin while many impurities remain unbound.
4. Sample Loading
With the column equilibrated and the sample prepared, the next step is loading.
- Reduce the flow rate during loading (compared with washing and elution) to enhance binding efficiency, especially on gravity‑flow or manual systems.
- Apply the sample either through an injection valve (in HPLC systems) or by carefully pipetting/connecting the sample to the top of the column.
- Allow the sample to enter the resin bed completely before resuming flow with Buffer A.
During this stage, the goal is to bind as much of the target as possible while allowing non‑interacting species to pass through. The flowthrough may be monitored or collected to verify that the target is retained.
5. Column Washing
After loading, the column is washed to remove unbound and weakly bound contaminants.
- Continue running Buffer A through the column, typically 3–5 (or more) column volumes, until the detector signal (for example, UV absorbance) returns to baseline.
- At this point, neutral and like‑charged molecules, and species with only weak interactions, should have been flushed out, leaving primarily the strongly bound target and closely related contaminants on the resin.
Good washing improves purity and ensures that the elution step focuses on the specific, strongly bound molecules.
6. Elution
Elution releases bound molecules from the resin by weakening electrostatic interactions, usually by increasing ionic strength or changing pH.
a. Salt Gradient Elution
A common strategy is to use a linear or stepwise gradient of increasing salt concentration.
- Start with 0% Buffer B (pure Buffer A) and gradually increase to a defined percentage (for example, 0–50% or 0–100% Buffer B over 10–20 column volumes).
- As the salt concentration rises, small ions (such as Na⁺ or Cl⁻) compete with bound molecules for charged sites on the resin, causing them to elute in order of increasing binding strength.
- Monitor the elution profile with a detector (for example, UV absorbance at 280 nm for proteins) and collect fractions across each peak.
Gradient elution gives detailed information about binding behavior and can be used to optimize separation conditions.
b. Step Elution
Once optimal conditions are known, many protocols use step elution to simplify and speed up purification.
- Instead of a continuous gradient, increase the salt concentration in one or several discrete steps (for example, jump from 0% to 20% Buffer B, then to 50% Buffer B).
- Each step is held for a few column volumes to elute a group of molecules with similar binding strength.
- Step elution is often used in routine production runs after the gradient conditions have been established.
c. pH‑Based Elution
In some cases, pH shifts are used instead of, or in addition to, salt gradients.
- Adjusting pH toward the isoelectric point of the bound molecule reduces its net charge and weakens the binding interaction.
- pH steps must be chosen carefully to avoid denaturation and to maintain compatibility with the resin and system.
Regardless of the elution mode, fractions are analyzed (for example, by UV, SDS‑PAGE, or activity assays) to identify those containing the purified target.
7. Final Column Washing
After elution of the target, it is common to perform a high‑salt wash.
- Run a buffer containing high salt (for example, 1–2 M NaCl) through the column for several column volumes.
- This step removes tightly bound impurities and helps prevent gradual buildup of contaminants on the resin.
A brief return to Buffer A can follow to check that the baseline is stable before regeneration or storage.
8. Column Regeneration and Storage
Regeneration restores the resin to a reproducible starting state and extends column lifetime.
- Apply a strong salt solution and/or specific cleaning agents recommended by the manufacturer to strip any remaining bound material.
- After cleaning, re‑equilibrate the column with several column volumes of Buffer A or a designated storage buffer.
- For long‑term storage, many protocols recommend a preservative such as 20% ethanol in water to prevent microbial growth, keeping the column well sealed.
Before the next run, the column should again be equilibrated in the starting buffer for the new method.
Mechanism at the Molecular Level
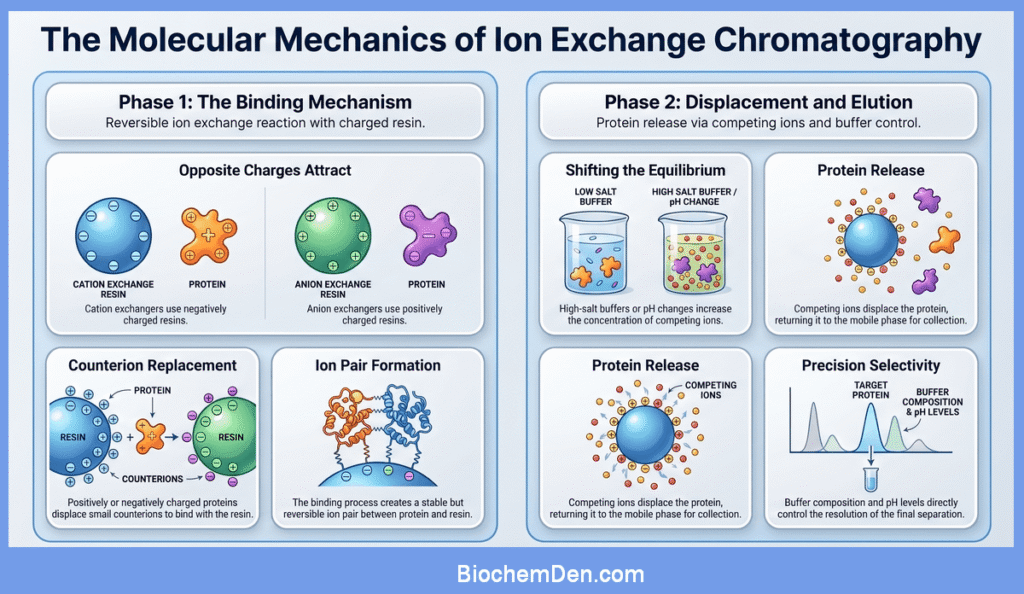
At the molecular level, ion exchange chromatography is governed by reversible ion exchange reactions. In a cation exchanger, the resin bears negatively charged groups and is initially balanced by small counterions such as sodium or hydrogen.
When a positively charged protein passes through the column, it can replace these small cations and bind to the resin, forming a new ion pair.
As a high-salt buffer or different pH is applied, the concentration or charge state of competing ions increases, and the equilibrium shifts toward the protein returning to the mobile phase.
In anion exchange chromatography, an analogous process occurs with negatively charged molecules competing with anions originally associated with positively charged resin groups.
This interplay between binding and displacement, controlled by buffer composition and pH, underlies the selectivity and resolution achievable with ion exchange chromatography.
Applications
Ion exchange chromatography is used in many areas of life science and analytical chemistry.
- Protein and peptide purification: Ion exchange chromatography is one of the most common techniques for purifying proteins and peptides based on differences in surface charge. It is frequently used for initial capture, intermediate purification, and final polishing of biomolecules.
- Nucleic acid purification: Anion exchange columns are used to purify plasmid DNA, genomic DNA, and RNA, taking advantage of their negatively charged phosphate backbone.
- Biopharmaceutical production: In bioprocessing, ion exchange steps are integrated into multistep purification processes to remove impurities, aggregates, and charge variants from therapeutic proteins.
- Analysis of inorganic ions: Ion exchange approaches are applied for the determination of inorganic cations and anions in water, environmental samples, and process streams.
- Water softening and deionization: Strong acid cation and strong base anion resins are used in water treatment to remove hardness and dissolved salts, producing softened or deionized water.
These applications illustrate how the same fundamental principle can be adapted to many different types of charged molecules and scales of operation.
Advantages and Limitations
Understanding the strengths and limitations of ion exchange chromatography helps in choosing and optimizing it for different tasks.
Advantages of Ion Exchange Chromatography
- High resolving power for molecules with subtle differences in charge and surface properties.
- Large sample‑handling capacity, suitable for both analytical and preparative‑scale separations.
- Broad applicability to proteins, peptides, nucleic acids, and small ions.
- Typically performed under mild, aqueous conditions that preserve the activity and structure of biomolecules.
- Can be integrated at multiple stages of a purification workflow, from capture to polishing.
Limitations of Ion Exchange Chromatography
- Requires careful control of pH and ionic strength, because small changes can significantly alter retention behavior.
- Method development can be time‑consuming, especially when optimizing gradients and buffer compositions.
- Strongly bound impurities may require rigorous cleaning procedures, which can shorten column lifetime if not managed properly.
Despite these limitations, ion exchange chromatography remains a versatile and reliable tool for separating charged molecules.
Typical Examples
A few typical examples help to connect the theory with practice.
- Separation of basic proteins on a cation exchange column, where proteins with different isoelectric points elute at different salt concentrations.
- Purification of negatively charged enzymes or DNA using DEAE‑cellulose anion exchange columns.
- Removal of trace ionic contaminants from pharmaceutical formulations using small‑scale ion exchange cartridges.
The chromatograms generated in these experiments show distinct peaks corresponding to individual components, illustrating how ion exchange chromatography resolves complex mixtures into their constituents.
Key Takeaways
Ion exchange chromatography separates ionizable molecules based on differences in net charge using charged resins and controlled buffer conditions.
By choosing between cation and anion exchangers, adjusting pH, and designing suitable salt gradients, it is possible to achieve high‑resolution separations for a wide variety of biomolecules and inorganic ions.
Discover more from Biochemistry Den
Subscribe to get the latest posts sent to your email.