Understanding the basics of pH is essential in biochemistry, chemistry, environmental science, and even everyday life.
The nature of any solution—whether it is acidic, alkaline (basic), or neutral—is determined by the concentration of hydrogen ions (H⁺) or hydroxyl ions (OH⁻) present in it.
The basics of pH refer to the measurement of hydrogen ion concentration used to determine whether a solution is acidic, neutral, or alkaline on a scale of 0 to 14.
This concept forms the foundation of the pH scale, which is widely used to measure acidity and alkalinity in laboratory experiments, biological systems, water quality testing, and industrial processes.
In 1909, the Danish biochemist Sørensen introduced a simple and effective method for expressing hydrogen ion concentration using a logarithmic scale, known as pH.
In this term, “p” stands for “power,” and “H” represents the hydrogen ion concentration.
This innovation made it easier to work with very small concentrations, which are common in chemical and biological systems.
“The negative logarithm of hydrogen ion concentration (in moles/liter)”
Thus,
1
PH = log ————– = – log [H⁺]
[H+]
Here, the symbol “p” denotes the negative logarithm, and [H⁺] represents the molar concentration of hydrogen ions.
This logarithmic relationship means that even small changes in pH correspond to significant changes in acidity or alkalinity, making pH a critical parameter in biochemical reactions, enzyme activity, and physiological balance.
For a perfectly neutral solution at 25°C (298 K), the concentration of hydrogen ions is 1.0 × 10⁻⁷ M. Substituting this value into the formula:
1
PH = log ——————— = log (1.0 X 107)
1.0 X 10⁻⁷
= log 1.0 + log 107
= 0 + 7.0
= 7.0
Thus, a pH of 7 is considered neutral, while values below 7 indicate acidity and values above 7 indicate alkalinity.
This value is not arbitrary—it arises from the ion product of water (Kw), a fundamental constant in chemistry that governs the equilibrium between hydrogen and hydroxyl ions in pure water.
For a standardized scientific explanation, refer to the official definition of pH provided by IUPAC.
Understanding the importance of pH goes far beyond theory. It plays a vital role in maintaining blood pH balance, soil fertility, food preservation, water safety, and industrial processes.
Even slight deviations in pH can significantly affect chemical reactions and biological systems, making it one of the most important concepts in both science and real-world applications.
Understanding the basics of pH is crucial for accurately interpreting chemical reactions, biological systems, and real-world applications.
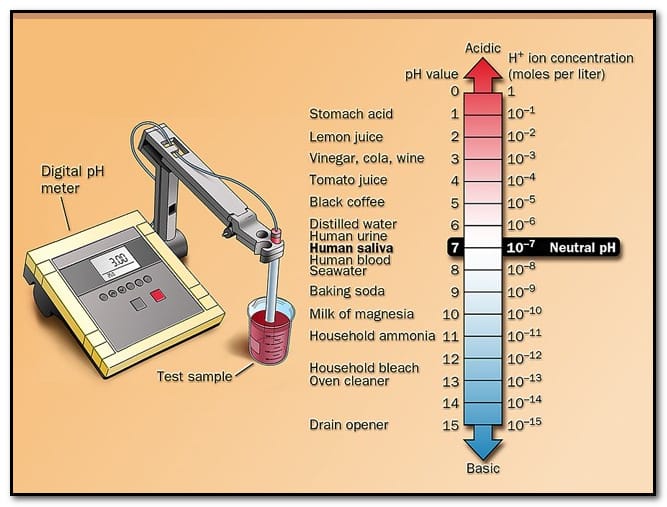
Basics of pH Scale with Examples (0–14)
The pH scale ranges from 0 to 14 and is used to measure how acidic or alkaline a substance is. Values below 7 indicate acidity, 7 is neutral, and values above 7 indicate alkalinity.
| pH Value | Nature | Common Examples |
|---|---|---|
| 0–1 | Strong Acid | Battery acid |
| 2 | Acidic | Lemon juice |
| 3 | Acidic | Vinegar |
| 4–5 | Weak Acid | Tomato juice, black coffee |
| 6 | Slight Acid | Milk |
| 7 | Neutral | Pure water |
| 7.4 | Slight Base | Human blood |
| 8–9 | Weak Base | Baking soda |
| 10–11 | Basic | Soap solution |
| 12–13 | Strong Base | Household ammonia |
| 14 | Strong Base | Sodium hydroxide |
This scale helps in understanding the basics of pH and how different substances behave in daily life and laboratory conditions.
Basics of pH in Real-Life Examples and Applications
The basics of pH can be easily understood through everyday examples:
- Food and Beverages: Citrus fruits like lemons are acidic, while milk is slightly acidic and safe for consumption.
- Human Body: Blood maintains a tightly regulated pH of about 7.35–7.45, essential for survival.
- Soil pH: Soil acidity or alkalinity determines nutrient availability and crop productivity in agriculture.
- Water Quality: Safe drinking water typically has a pH between 6.5 and 8.5, ensuring it is neither too acidic nor too alkaline.
These examples highlight how pH influences health, the environment, and daily life.
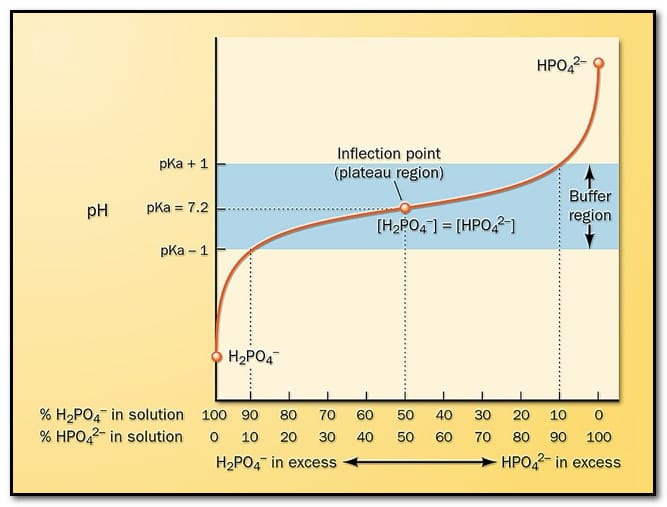
Buffer Systems in the Basics of pH and Their Role
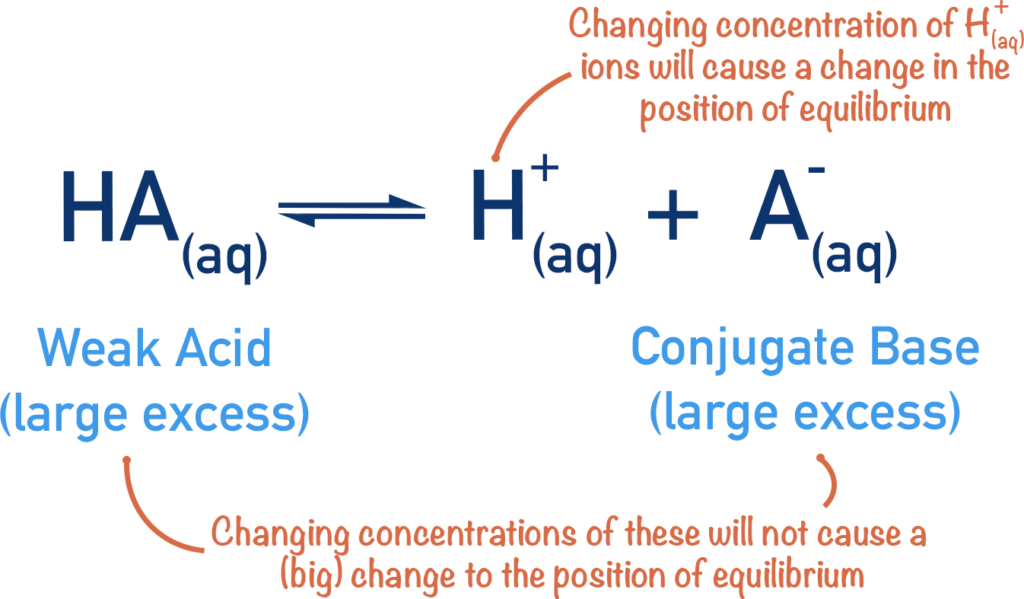
Buffer systems are solutions that resist sudden changes in pH when small amounts of acids or bases are added. They play a crucial role in maintaining stable pH levels in biological and chemical systems.
- Buffers typically consist of a weak acid and its conjugate base.
- The most important biological buffer is the bicarbonate buffer system, which helps maintain blood pH.
- Buffers prevent drastic pH changes that could disrupt enzyme activity and metabolic processes.
Understanding buffer systems is an important part of the basics of pH, especially in biochemistry and physiology.
Importance of pH in Chemistry and Biology
The basics of pH help explain why maintaining the correct pH level is critical for agriculture, human health, and industrial processes.
The importance of pH extends across various fields, including agriculture, biology, medicine, environmental science, and industrial applications.
The role of pH in ecosystems is further explained through its impact on water systems in this guide on pH and water quality importance.
The pH level of a solution directly influences chemical reactions, biological processes, and overall system stability.
- Soil pH for plant growth: Different crops require a specific soil pH range for optimal nutrient absorption, microbial activity, and improved crop yield. For example, slightly acidic soils (pH 6–7) are ideal for most crops.
- Industrial and biochemical processes: Maintaining a precise optimal pH level is crucial in industrial manufacturing, fermentation, pharmaceutical production, and enzyme reactions, where even slight pH changes can significantly impact efficiency and product quality.
- Human body and blood pH balance: The human body tightly regulates blood pH (around 7.35–7.45). Any imbalance can lead to serious conditions such as acidosis or alkalosis, highlighting the critical role of pH in homeostasis.
- In chemical analysis, pH is a crucial parameter that influences titrations, buffer systems, and reaction mechanisms. Accurate pH measurement is essential for laboratory experiments and research.
- Environmental importance: pH determines water quality, the survival of aquatic life, and pollution levels, making it a crucial factor in environmental monitoring.
Development and Basics of pH Concept
- The pH scale was developed using pure water as the reference standard. It is based on the ionization of water into hydrogen ions (H⁺) and hydroxyl ions (OH⁻).
- Experimentally, it is observed that:
- Only 1 molecule out of 550,000,000 molecules of water ionizes into H⁺ and OH⁻ ions.
- This corresponds to approximately 1 gram of hydrogen ions in 10,000,000 liters of water.
- Therefore, 1 liter of pure water contains 1 × 10⁻⁷ grams (or moles) of H⁺ ions.
- To simplify working with such extremely small values, scientists introduced the concept of “power of hydrogen” (pH), where only the exponent (power) is used. This led to the development of the convenient and widely accepted logarithmic pH scale, making calculations and comparisons easier in biochemistry and chemistry.
These concepts form the basics of pH, making it easier to understand how scientists measure and express acidity and alkalinity.
Biological Significance of pH in Living Systems
The basics of pH are crucial for understanding how biological molecules function and maintain stability in living systems.
The biological significance of pH is profound, as it governs the structure, function, and interaction of biomolecules in living systems.
A) Tautomeric forms of purines and Pyrimidines
Tautomerization is a special type of isomerism in which a proton shifts within a molecule, accompanied by a rearrangement of bonds.
- Purines and pyrimidines exist in different tautomeric forms depending on pH.
- At physiological body pH (~7.4), the correct tautomeric forms are stabilized.
- These forms are essential for accurate hydrogen bonding in DNA and RNA base pairing.
Thus, pH plays a crucial role in maintaining the structural integrity and genetic stability of nucleic acids.
B) Isoelectric pH (pI)
pH has a significant impact on the ionization of biomolecules, including amino acids, proteins, nucleic acids, and phospholipids.
- At a specific pH, known as the isoelectric point (pI), molecules carry no net charge.
- At this stage, they exist as zwitterions (having both positive and negative charges).
- These molecules do not migrate in an electric field and tend to precipitate easily due to reduced electrostatic repulsion.
This concept is essential in protein purification and biochemical analysis.
C) Isolation of Proteins and Amino Acids
The charge of proteins and amino acids depends on the surrounding pH of the solution, which is exploited in separation techniques.
- Ion-exchange chromatography
- Paper electrophoresis
- Isoelectric focusing
These techniques rely on pH-dependent charge variations to separate, isolate, and purify biomolecules from complex biological samples. This is widely used in biotechnology, proteomics, and clinical diagnostics.
D) Optimum pH for Enzyme Activity
Every enzyme has an optimum pH at which it functions most efficiently.
- pH influences the ionization state of amino acid residues in enzymes.
- It affects ionic bonds and hydrogen bonds, which stabilize the three-dimensional protein structure.
- Changes in pH can lead to enzyme denaturation or reduced activity.
For example:
- Pepsin works best in acidic conditions (stomach).
- Trypsin functions optimally in alkaline conditions (intestine).
Thus, maintaining the correct pH is crucial for enzyme activity, metabolism, and overall physiological function.
E) pH and Enzyme Kinetics
The rate of enzyme-catalyzed reactions is highly dependent on pH.
- Each enzyme shows maximum activity at a specific optimum pH range.
- Deviations from this range can alter the active site structure, reducing substrate binding.
- Extreme pH conditions may lead to enzyme denaturation and complete loss of function.
This makes pH a critical factor in metabolic pathways and biochemical reactions.
F) pH and Oxygen Transport (Bohr Effect)
pH plays a key role in oxygen transport in blood through the Bohr effect.
- A decrease in pH (more acidic conditions) reduces hemoglobin’s affinity for oxygen.
- This promotes the release of oxygen to actively respiring tissues.
- An increase in pH enhances oxygen binding in the lungs.
Thus, pH helps regulate efficient oxygen delivery and respiration.
G) pH in Cellular Metabolism
The intracellular pH is tightly regulated to support normal cellular functions.
- It affects glycolysis, ATP production, and energy metabolism.
- Abnormal pH can disrupt cell signaling pathways and enzyme systems.
- Cells use buffer systems to maintain a stable internal pH environment.
Maintaining a proper pH balance is crucial for cell survival and optimal metabolic efficiency.
H) pH and Membrane Transport
The pH gradient across biological membranes is vital for transport mechanisms.
- It drives active transport and proton pumps (e.g., ATP synthase).
- Essential for processes like oxidative phosphorylation and photosynthesis.
- Maintains electrochemical gradients necessary for nutrient uptake.
This highlights the crucial role of pH in cellular energy production and transport processes.
I) pH and Drug Absorption
pH has a significant influence on drug absorption and pharmacokinetics.
- Drugs exist in both ionized and non-ionized forms, depending on the pH.
- Only the non-ionized form easily crosses cell membranes.
- The pH levels of the stomach and intestine determine the solubility and absorption rate of drugs.
This principle is crucial in drug design, delivery, and therapeutic effectiveness.
J) pH and Microbial Growth
The growth of microorganisms is strongly influenced by environmental pH.
- Different microbes thrive at different pH ranges (acidophiles, neutrophiles, alkaliphiles).
- pH affects enzyme systems, nutrient availability, and membrane integrity in microbes.
- Controlling pH is widely used in food preservation and fermentation technology.
Thus, pH is a key factor in microbiology, food science, and biotechnology.
Frequently Asked Questions (FAQs) on the Basics of pH
The basics of pH refer to the measurement of hydrogen ion concentration used to determine whether a solution is acidic, neutral, or alkaline
What are the basics of pH?
The basics of pH refer to measuring the hydrogen ion concentration in a solution to determine whether it is acidic, neutral, or alkaline. The pH scale ranges from 0 to 14, with 7 as neutral. Lower values indicate acidity, while higher values indicate alkalinity.
Why is pH important in biological systems?
pH is crucial in biological systems because it affects enzyme activity, protein structure, and metabolic reactions. Even slight changes in pH can disrupt cellular processes, making it essential for maintaining physiological balance, proper functioning of biomolecules, and overall homeostasis in living organisms.
How is pH calculated?
pH is calculated using the formula pH = –log [H⁺], where [H⁺] represents the hydrogen ion concentration in moles per liter. This logarithmic scale simplifies very small values, making it easier to measure and compare the acidity or alkalinity of different solutions.
What is the normal pH of pure water?
The pH of pure water at 25°C is 7, which is considered neutral. This value is based on the equal concentration of hydrogen ions (H⁺) and hydroxyl ions (OH⁻), derived from the ion product of water, making it a standard reference point in chemistry.
What is the role of pH in enzyme activity?
pH influences enzyme activity by affecting the ionization of amino acids at the active site. Each enzyme has an optimum pH where it works most efficiently. Outside this range, enzyme structure can change, leading to reduced activity or complete denaturation.
How does pH affect soil and plant growth?
Soil pH affects nutrient availability, microbial activity, and plant growth. Most crops grow best in slightly acidic to neutral soils (pH 6–7). Incorrect pH levels can limit nutrient absorption, reduce crop yield, and negatively impact overall agricultural productivity.
What is the isoelectric point (pI)?
The isoelectric point (pI) is the pH at which a molecule, such as a protein or amino acid, carries no net electrical charge. At this point, molecules exist as zwitterions and do not migrate in an electric field, making it important for protein purification techniques.
How does pH affect drug absorption?
pH affects drug absorption by determining whether a drug is in its ionized or non-ionized form. Non-ionized forms cross cell membranes more easily. The pH of the stomach and intestine influences drug solubility, absorption rate, and overall effectiveness in the body.
Final word
The basics of pH are central to understanding biochemistry, chemistry, and life sciences. The pH scale, based on hydrogen ion concentration, provides a straightforward yet powerful method for measuring acidity, alkalinity, and neutrality in solutions. Its logarithmic nature allows accurate representation of even very small changes in ion concentration, which can have significant chemical and biological effects.
A strong grasp of the basics of pH provides the foundation for understanding both chemical principles and biological processes.
The concept of pH is deeply connected to the ionization of water, buffer systems, and equilibrium reactions, making it essential for interpreting chemical behavior and analytical processes. It plays a key role in determining how substances interact, react, and maintain stability in different environments.
In biological systems, pH plays a crucial role in maintaining enzyme activity, protein structure, nucleic acid stability, and metabolic processes. It regulates important phenomena such as tautomerism of nitrogenous bases, isoelectric point of biomolecules, membrane transport, oxygen delivery, and cellular metabolism. Even slight deviations in pH can disrupt these processes, highlighting their importance in maintaining physiological balance and homeostasis.
Beyond biology, pH has numerous applications in agriculture (soil pH and crop productivity), environmental science (water quality and pollution control), medicine (regulation of blood pH and drug absorption), and industry (fermentation, pharmaceuticals, and chemical manufacturing).
Overall, a clear understanding of pH provides a strong foundation for exploring chemical reactions, biological systems, and real-world applications, making it one of the most essential concepts in science.
Discover more from Biochemistry Den
Subscribe to get the latest posts sent to your email.